Research
|
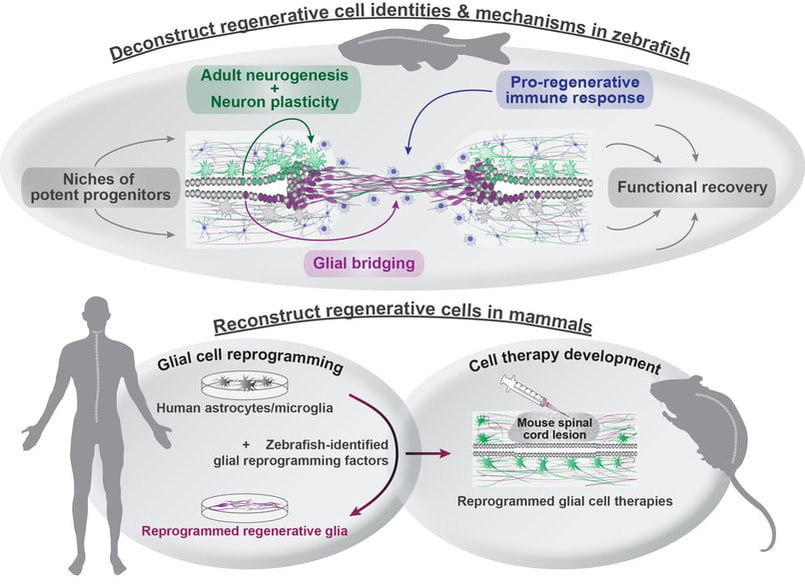
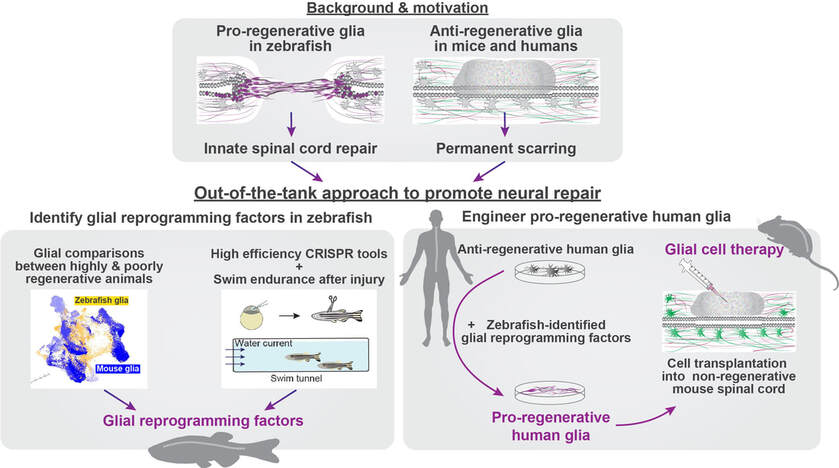
OVERALL GOAL & MOTIVATION Our long-standing goal is to elucidate evolutionarily conserved mechanisms of spinal cord regeneration, and to develop zebrafish-inspired interventions to promote neural repair in mammals. Adult zebrafish possess an elevated regenerative capacity and lack the anti-regenerative complications displayed after mammalian nervous system injuries. Our laboratory aims to leverage the strengths of the zebrafish model system to uncover pro-regenerative cell identities and mechanisms in highly regenerative vertebrates, and to reconstruct analogous mechanisms in poorly regenerative mammals. |
SPECIFIC PROJECTS
I- POTENCY AND CONTRIBUTION OF SPINAL CORD PROGENITORS
I- POTENCY AND CONTRIBUTION OF SPINAL CORD PROGENITORS
|
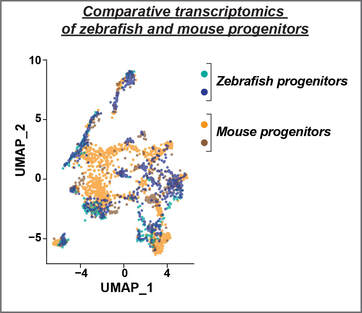
We hypothesize that localized niches of lineage-restricted progenitors contribute to either glial or neuronal regeneration after injury. We are pursuing a combination of cellular and molecular approaches to test this hypothesis. Cellularly, we are using genetic lineage tracing to dissect the contributions of progenitor cells during regeneration. Molecularly, we are using single-cell trancriptomics to determine progenitor cell identities in the zebrafish and mouse spinal cord. These studies provide a discovery platform to understand the molecular and cellular basis of regenerative capacity across species.
|
II- REGENERATIVE GLIAL CELL IDENTITIES AND MECHANISMS
|
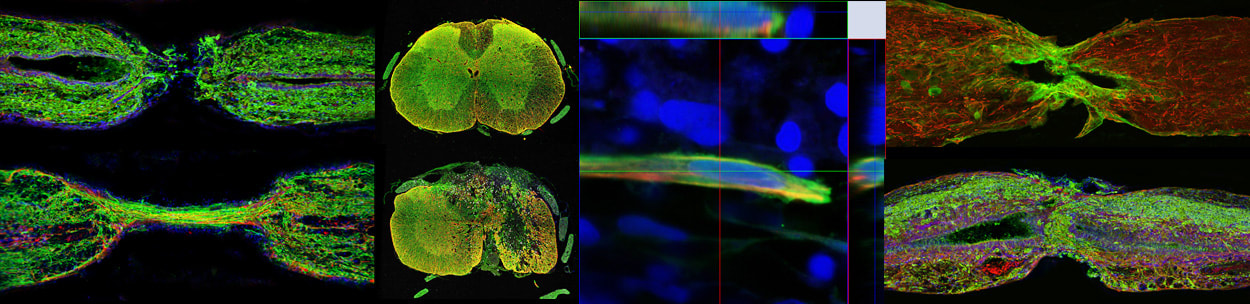
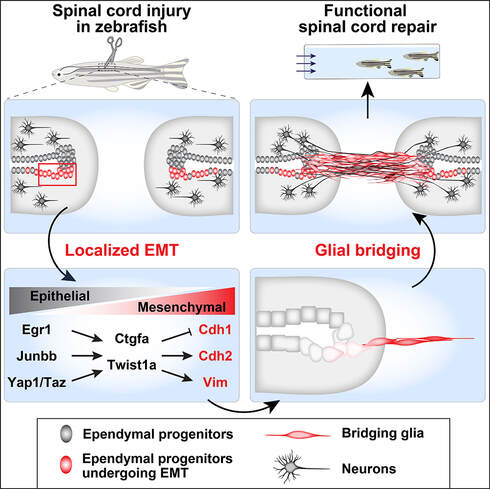
In zebrafish, specialized bridging glia connect the transected spinal cord and support axon regrowth across the lesion. Using bulk and single-cell sequencing, we found that pro-regenerative zebrafish glia activate an epithelial-to-mesenchymal transition (EMT) gene program and that EMT gene expression distinguishes mammalian astrocytes from zebrafish bridging glia. Functionally, we found that localized niches of glial progenitors undergo EMT after spinal cord injury in zebrafish and, using large-scale CRISPR/Cas9 mutagenesis, we identified a gene regulatory network that activates EMT and drives functional regeneration (Klatt Shaw*, Muraleedharan Saraswathy*, Zhou* et al., Dev Cell, 2021; Klatt Shaw and Mokalled, G3; 2021). These studies reveal that non-regenerative mammalian glia lack an essential EMT-driving gene regulatory network that reprograms pro-regenerative zebrafish glia after injury.
|
III- ADULT NEUROGENESIS MECHANISMS
|
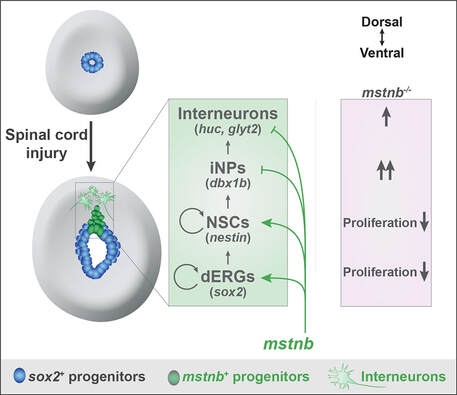
Studies of innate spinal cord regeneration have focused on glial and axonal bridging as primary repair mechanisms. However, how local neurogenesis impacts functional recovery is unknown. Using a candidate approach, we uncovered dynamic expression of myostatin b (mstnb) in a niche of dorsal ependymal progenitors after injury, and found that mstnb is a negative regulator of neurogenesis (Muraleedharan Saraswathy*, Zhou* et al., bioRxiv, 2021). We are currently using unbiased single-cell transcriptomics and CRISPR/Cas9 screening to dissect neurogenesis mechanisms in adult zebrafish. These studies establish the importance of local neurogenesis for functional spinal cord repair.
|
IV- DISCOVERY PROJECTS
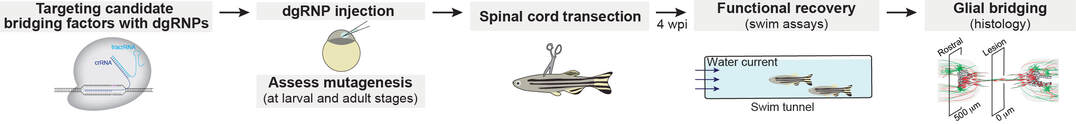
We recently described an experimental pipeline that combines high-efficiency CRISPR/Cas9 mutagenesis with functional phenotypic screening to identify genes required for spinal cord repair in adult zebrafish (Burris et al., JoVE, 2021; Klatt Shaw and Mokalled, G3, 2021). These studies provide a platform that enabled us to perform medium- to large-scale genetic studies in adult zebrafish.
V- DEVELOPING ZEBRAFISH-INSPIRED HUMAN CELL THERAPIES
As recent years brought major advances in neuron-intrinsic regenerative approaches, we propose that providing a permissive glial environment for regenerating neurons is an upcoming challenge and an underexplored avenue in regenerative medicine. We are using comparative transcriptomics to comprehensively define pro-regenerative cell identities and states in zebrafish, and to identify molecular pathways that distinguish pro-regenerative cells in zebrafish from non-regenerative cells in mammals. Using the power of cell fate reprogramming, we are engineering human glial cells that mimic the transcriptional and functional profiles of pro-regenerative cells in zebrafish. We posit that shifting human glial cell states towards pro-regenerative phenotypes will promote debris clearance, counteract and minimize scarring, and provide natural scaffolds for neural regeneration.